Researchers have mixed nanodiamonds with gutta-percha to create a new material that is stronger than the original and has antimicrobial capabilities. The nanodiamond gutta-percha may be able to prevent reinfection after root canal therapy, according to the authors of a new study.
 Dean Ho, PhD, is a professor at the UCLA School of Dentistry and co-director of the Weintraub Center for Reconstructive Biotechnology at UCLA. Image courtesy of UCLA.
Dean Ho, PhD, is a professor at the UCLA School of Dentistry and co-director of the Weintraub Center for Reconstructive Biotechnology at UCLA. Image courtesy of UCLA.In the study, which was published in ACS Nano (October 9, 2015), researchers from the University of California, Los Angeles (UCLA) bound amoxicillin to nanodiamonds, then incorporated the nanodiamonds into standard gutta-percha that is often used for root canal therapy. The new rubber material may one day allow clinicians to directly target an infection without affecting the rest of a patient.
"We found that when the bacteria touch or make contact with this rubber, it can block bacterial growth because it's a contact-based drug delivery," said Dean Ho, PhD, one of the researchers and a professor at the UCLA School of Dentistry, in an interview with DrBicuspid.com. "And it worked. It worked beautifully. You can still x-ray it, which the clinicians need. So you get the best of all worlds. Increased strength, the ability to hold a drug or more than one down the road, it's not expensive, and it can be readily adapted to existing clinical protocols."
Nanodiamonds bind and strengthen
Nanodiamonds are like soccer balls with sharp edges and a unique charge, making them ideal for bonding with all types of drugs, Dr. Ho explained. They are also a sustainable nanomedicine, because they are a byproduct of mining and refining operations. Unlike the bright, shiny diamonds we typically think of, nanodiamonds are similar to dirt and can be cheaply attained and processed for biomedicine.
Dr. Ho, who is also co-director of the Weintraub Center for Reconstructive Biotechnology at UCLA, became interested in using nanodiamonds to improve root canal therapies after getting one himself. When Dr. Ho asked his oral surgeon about the shortcomings of gutta-percha, his clinician explained that it could be stronger and that it also cannot protect the tooth against any remaining bacteria, risking that the treated tooth may become infected again.
Dr. Ho realized that it may be possible to use nanodiamonds to create an improved version of gutta-percha, so his team got to work. First, they loaded amoxicillin onto the nanodiamonds and waited five to seven days for it to incorporate. Then, the team used sonication (sounds waves) to add the nanodiamonds to the gutta-percha matrix.
Once the nanodiamond gutta-percha was created, two dental students placed the material into the root canals of extracted human teeth, just as if they were performing the procedure on a patient. Dr. Ho and his team tested the antimicrobial efficiency, radiopacity, stress, and strain of material to see how it compared with unmodified gutta-percha.
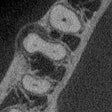
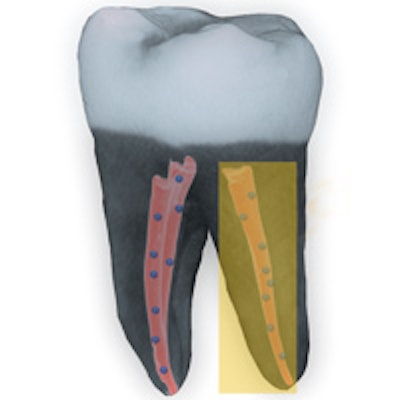
 A 3D image of a tooth filled with nanodiamond-enhanced gutta-percha and an individual gutta-percha point. Image courtesy of American Chemical Society/Dong-Keun Lee.
A 3D image of a tooth filled with nanodiamond-enhanced gutta-percha and an individual gutta-percha point. Image courtesy of American Chemical Society/Dong-Keun Lee.They found that the nanodiamond gutta-percha was able to kill a significant amount of bacteria, which was not true for the unmodified gutta-percha. They also noted that, despite the modified material being generally stronger than the unmodified gutta-percha, they found no difference in the radiographic processes. The new material may be able to increase the success rate for root canal therapy by preventing reinfection of the tooth, the authors concluded.
"It really surprised us that there were no apparent differences between the two, which was surprisingly wonderful because that speaks volumes to the clinical implications," Dr. Ho said. "Now we can really focus on making sure this is ready for the clinic."
Clinical trial
“Nanomedicine and bioengineering are areas that can drastically impact the field of dental health.”
Dr. Ho's team is currently designing a one- to two-year clinical trial to evaluate the durability of the nanodiamond gutta-percha and also its ability to prevent infection. Also, one of the first clinicians to use the nanodiamond gutta-percha on humans will be the oral surgeon who helped inspire the creation of the material in the first place.
Dr. Ho said he is excited for the next steps, and, if all goes well, the material may be ready for nonclinical trial use within five years. He also believes that this is just the start for nanodiamonds and new technologies in dentistry.
"Nanomedicine and bioengineering are areas that can drastically impact the field of dental health and that also goes into training of the next generation of leadership," he said. "It's really exciting to work with dental clinicians that are very dynamic and very versatile with regards to innovating alongside us. That mindset of being progressive and open to adopting new technologies is going to lead to some exciting new developments in dental health."