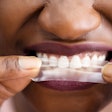
Attorneys have requested a delay in a consolidated lawsuit filed against GlaxoSmithKline (GSK) and Procter & Gamble that alleges the zinc in these denture adhesives can cause numerous health problems and that the manufacturers failed to properly warn consumers about these risks, according to a story on aboutlawsuits.com.
The request, which was made December 1, asks the U.S. District Court for the Southern District of Florida to extend the deadline for initial discovery established in the federal multidistrict litigation.
The discovery and depositions have not yet been completed due to the "complex scheduling involving a large number of counsel and witnesses," according to the story.
The complaints all contain similar allegations that Procter & Gamble, which manufactures Fixodent, and GSK, which manufactures Poligrip, failed to warn consumers that their products contain zinc, which may enter the body at high levels when the denture cream is used. The plaintiffs allege that they suffered severe and permanent neurological damage from denture cream zinc poisoning.
In August, GSK reported that it had reached settlement agreements in the vast majority of Poligrip cases filed against the company. Procter & Gamble has vowed to fight the lawsuits, and Judge Cecilia Altonaga has scheduled the first Fixodent trial to begin on June 6, 2011.
GSK contends that the products are safe when used as directed, but that some consumers apply more adhesive than directed and use it more than once per day.
Even so, the company announced earlier this year that it would stop manufacturing Poligrip products and replace them with zinc-free alternatives. In an advisory issued February 18, the company warned consumers about the potential health risks associated with long-term excessive use of zinc-containing denture adhesives, including Super Poligrip Original, Super Poligrip Ultra Fresh, and Super Poligrip Extra Care.
Copyright © 2010 DrBicuspid.com